Valueable properties of aluminium
Explore the valuable properties of aluminium, including its low density, high corrosion resistance, and impressive thermal and electrical conductivity.
Combination of aluminium properties
Low density
- Aluminum – is one of the most light industrial construction metal.
- Density of aluminium approximately three times lower, than steel or copper.
- This gives high specific strength – strength per unit mass.
 Figure 1 – Aluminium Volume per Unit Weight [3]
Figure 1 – Aluminium Volume per Unit Weight [3]

Figure 2 – The Effect of Alloying Elements on Tensile Strength, hardness, Impact Sensibility and Ductility [3]
 Figure 3 – Density-Related Strength of Aluminium in Comprasion with Various Metals and Alloys [3]
Figure 3 – Density-Related Strength of Aluminium in Comprasion with Various Metals and Alloys [3]
 Figure 4 – Stress-Strain Curves of Aluminium in Comparision with Various Metals and Alloys [3]
Figure 4 – Stress-Strain Curves of Aluminium in Comparision with Various Metals and Alloys [3]
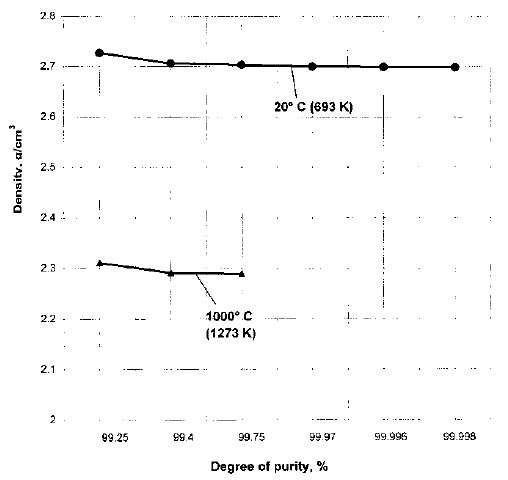 Figure 5 – The Effects of Aluminium Purity and Temperature on Aluminium Density [2]
Figure 5 – The Effects of Aluminium Purity and Temperature on Aluminium Density [2]
Сorrosion resistance
High corrosion resistance of aluminium may be one of its most valuable properties. Aluminium has a high corrosion resistance due to a thin layer of aluminum oxide on its surface,. This oxide film is formed instantly, as soon as a fresh aluminium surface is brought into contact with air. In many cases, this property allows the use of aluminium without any special surface treatment. If you need additional protective or decorative coating, the applied anodizing or painting the surface.
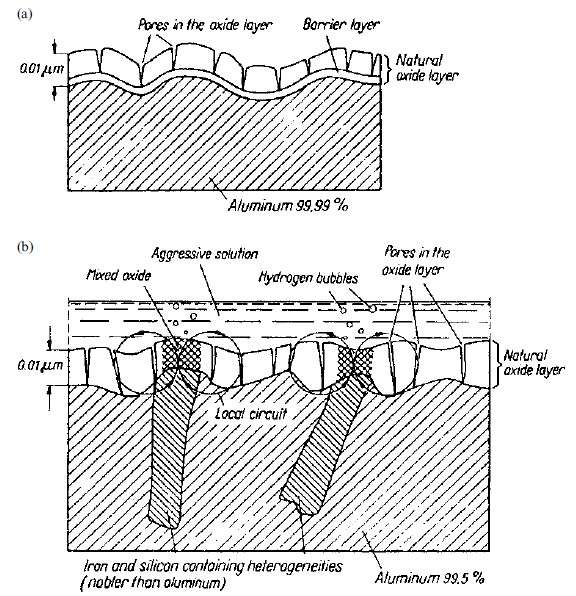 Figure 6
Figure 6
a – natural oxide film on ultra-pure aluminium;
b – corrosion of aluminium with a purity of 99,5% with a natural oxide film
in a corrosion-aggressive environment [2]
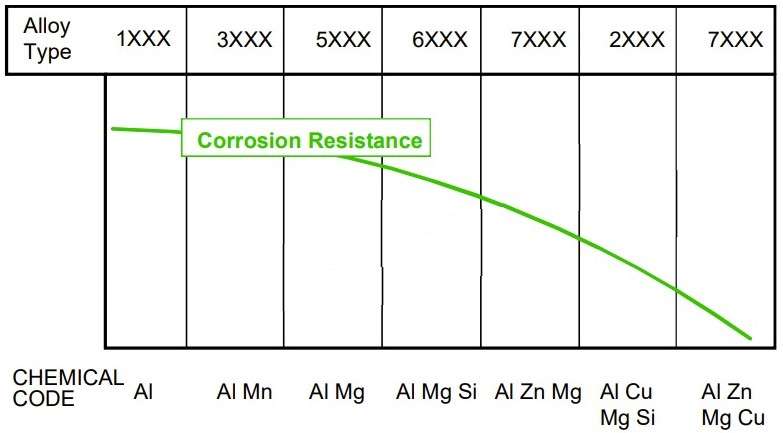
Figure 7 – Effects of Alloying Elements on Corrosion Resistance and Fatigue Strength of Aluminium Alloys [3]
 Figure 8 – Pitting Corrosion Behaviour of 3103 Mill Finish Aluminium Sheet [3]
Figure 8 – Pitting Corrosion Behaviour of 3103 Mill Finish Aluminium Sheet [3]
Strength
The strength properties of pure aluminium are rather low (figure 9). Mechanical properties can increase:
- when adding alloying elements
- thermal hardening (Figure 10)
- strain hardening (Figure 11).
Typical alloying elements are:
- manganese,
- silicon,
- copper,
- magnesium,
- zinc.

Figure 9 – Influence of purity of aluminum on its strength and hardness [2]

Figure 10 – Mechanical properties of high-purity wrought
aluminum-copper alloys in various states [2]
(O – annealed, W – right off after quenching, T4 – naturally aged, T6 – artificially aged)
 Figure 11 – Mechanical properties of aluminum 99,50 %
Figure 11 – Mechanical properties of aluminum 99,50 %
depending on the degree of cold deformation obtained [2]
Durability at low temperatures
- Steel becomes brittle at low temperatures.
- Aluminum at low temperatures increases its strength and maintains high toughness.
- This property makes it possible to use aluminum in space cold conditions.
 Figure 12 – Tensile Properties of 6061 Alloy Heat Treated, Artifically Aged [3]
Figure 12 – Tensile Properties of 6061 Alloy Heat Treated, Artifically Aged [3]
Thermal conductivity
Aluminium conducts heat three times faster, than steel. This physical property is very important in heat exchangers to heat or cool the working environment. From here is the extensive use of aluminum and its alloys in cookware, air conditioners, primyshlennom and automotive heat exchangers.
 Figure 13 – Thermal Conductivity of Aluminium
Figure 13 – Thermal Conductivity of Aluminium
Compared with other Metals [3]
Reflectivity
Aluminium is an excellent reflector of radiant energy over the entire range of wavelengths. This physical property makes it possible to use it in devices, which work against ultraviolet spectrum through the visible spectrum to the infrared spectrum and thermal waves, as well as such electromagnetic waves, like radio waves and radar waves [1].
Aluminum has the ability to reflect more 80 % light waves, which provides it with widespread use in lighting devices (figure 14). Due to the physical properties, it is used in thermal insulation materials. for instance, aluminium roofing reflects a large proportion of the solar radiation, that provides indoor air cool in the summer and, in the same time, It retains heat the room in winter.

Figure 14– Reflectivity of Aluminium [2]
 Figure 15 – Reflectivity and Emissivity of Aluminium
Figure 15 – Reflectivity and Emissivity of Aluminium
with Various Surface Treatments [3]
 Figure 16 – Comparison of Reflectivity of Various Metals [3]
Figure 16 – Comparison of Reflectivity of Various Metals [3]
Electrical properties
- Aluminium is one of the two available metal, which have sufficiently high electrical conductivity, to apply them as electrical conductors.
- Electrical conductivity of aluminium 1350 is about 62 % from the international standard IACS, that is the electrical conductivity of annealed copper.
- However, the specific weight of aluminum is only one third of the specific weight of copper. It means, that he spends twice as much electricity, than copper of the same weight.
- This physical property allows aluminium to be widely used in high voltage power lines, transformer, electric buses and socket light bulbs.
 Figure 17 – Electrical Properties of Aluminium [3]
Figure 17 – Electrical Properties of Aluminium [3]
Magnetic properties
- Aluminium has the property not to be magnetized in electromagnetic fields.
- This makes it useful in protecting the equipment from exposure to electromagnetic fields.
- Another application of this feature is computer discs and parabolic antenna.
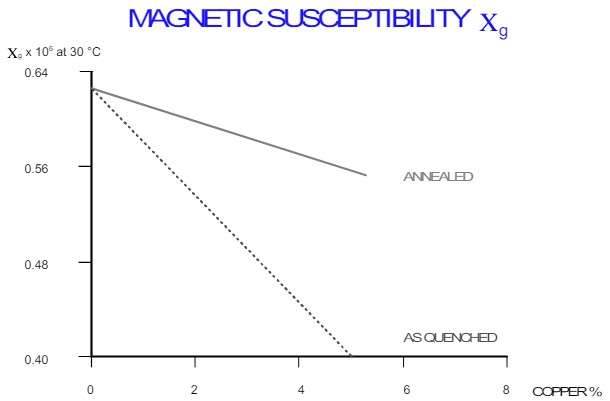 Figure 18 – Magnetic Susceptibility of AlCu Alloy [3]
Figure 18 – Magnetic Susceptibility of AlCu Alloy [3]
No toxic properties
This property of aluminium – no toxicity – was discovered at the beginning of its industrial development. It is this property of aluminum has enabled its use for the manufacture of kitchen utensils and appliances, without any harmful effects to the human body. Aluminium with its smooth surface is easy to clean, it is important to ensure high hygiene when cooking. Aluminium foil and containers widely and safely used in the packaging of a direct contact with food.
Sound insulation properties
This property makes aluminium a use when performing acoustic ceilings.
The ability to absorb impact energy
Aluminium has a modulus three times smaller, than steel. This physical property makes it a great advantage for the manufacture of automobile bumpers and other car security funds.
Figure 19 – Automotive aluminum profiles
to absorb impact energy in an accident
Fireproof properties
- Aluminium pieces forms no sparks upon impact against each other, as well as other non-ferrous metal. This is important for use with increased fire safety measures for structures.
- At the same time, with increasing temperature above 100 degrees Celsius, the strength of aluminium alloys is significantly reduced (Figure 20).
 Figure 20 – Tensile Strength of 2014-T6 at Test Temperatures [3]
Figure 20 – Tensile Strength of 2014-T6 at Test Temperatures [3]
Technological properties
Ease, with which the aluminium can be processed into any form – workability, It is one of the most important of its advantages. Very often it can successfully compete with the cheaper materials, which are much more difficult to handle:
- This metal may be casting by any method, which is known to metallurgists.
- It can be rolled up to thicknesses of up to foil, thinner sheets of paper which.
- Aluminium plates can be stamped, It extends, planted and mold all known methods of metal forming.
- Aluminium can be forged by all means of forging
- Aluminium wire, dragged from a round bar, It may then be woven into electric cables of any type and size.
- Almost no shape restrictions extruded profiles, in which it is prepared from this metal extrusion.
Sources:
- Aluminium and Aluminium Alloys. – ASM International, 1993.
- A. Sverdlin Properties of Pure Aluminum // Handbook of Aluminum, Vol. 1 /ed. G.E. Totten, D.S. MacKenzie, 2003
- TALAT Lecture 1501- Aluminium:Physical Properties, Characteristics and Alloys
- TALAT Lecture 3710 – Case Study on Can Making /Eric Wootton, Alcan Deutschland GmbH, Göttingen – 1994

