Aluminium melting dross
Aluminium melting dross is the surface layer on the aluminium melt, which has a high oxide content.
This aluminium melting dross:
- formed on the surface of aluminium melts as a result of interaction aluminium with air gases, primarily, with oxygen
- is a physical mixture of oxides and other solid compounds, as well as molten aluminium, which is inside them
- is an inevitable by-product [1].
What is aluminium melting dross
Aluminium melting dross is a source of problems in all industries, who are engaged in melting aluminium and aluminium alloys.
- The dross is a valuable by-product, which greatly depends on the profitability of production operations.
- This dross, in addition to oxides, often contains a large amount of usable metallic aluminum, which is captured by these oxides (Figure 1).
- If proper measures are not taken to prevent excessive dross formation, as well as maximum extraction of metal aluminum from it, then this can lead to significant losses of potentially usable metal.
 Figure 1 – The aluminium dross with high aluminium content
Figure 1 – The aluminium dross with high aluminium content
under an electron microscope [2]
Influence of melting technology
The most aluminium is melted in gas-fuel furnaces (Figure 2). These furnaces are characterized by the following technological factors, which have a great influence on the technological losses of aluminium in the furnace.
 Figure 2 – Cross section reverberatory melting furnace:
Figure 2 – Cross section reverberatory melting furnace:
1) transfer of heat to the surface of the melt by radiation;
2) heat transfer deep into the melt by convection [3]
Gas / air ratio on gas burners
The first factor is the gas/air ratio on gas burners. Excessive air supply leads to an increased rate of dross formation, if a fresh aluminum surface appears as a result of any movement of the melt surface. The situation is getting worse, if the burners hits directly on the surface of the melt or on the aluminium charge.
Melt holding temperature
The second important factor is the holding temperature of the aluminium melt. Temperature rise only by 25-55 ºС can have a very large impact (Figure 3). 
Figure 3 – Logarithmic increase in the rate of aluminum oxidation
with increasing holding temperature (in percent) [1]
Need to keep in mind, that thermocouples in a bath of molten aluminum actually measure the temperature in the depth of the bath. It is impossible to measure the temperature directly on the uppermost surface. It can be significantly hotter, than the temperature in the depth of the melt. Consequently, heat, shown in Figure 1, can be achieved at the surface of the melt. This will give a sharp increase in aluminum oxidation and will lead to an increase in the thickness of the dross layer.
Dross layer thickness
Excessive thickness of the dross layer above the melt can have a thermal insulating effect and cause increased burner operation., to maintain the desired melt temperature. In this case, the temperature in the dross layer rises and, respectively, the rate of metal oxidation increases, which enters the dross from the surface of the melt. As a result, there is a logarithmic increase in the loss of metal during melting.. The effect of increasing the dross thickness on the melting rate is shown in the Figure 4, which is confirmed by most aluminum smelters.
- For this reason, in aluminium melting furnaces, the thickness of the dross on the surface of the melt is usually not allowed to exceed 40 mm.
Figure 4 – Influence of the thickness of the dross layer on the reduction of the melting rate of the reverberatory melting furnace (in percent) [1]
Usually the temperature of the dross layer is about 50-85 ºС above, than the melt temperature. Therefore, the high temperature of the melt causes the risk of ignition of the dross and spontaneous combustion of metallic aluminium.. This thermite reaction can have serious consequences.. If we allow the development of this reaction, the temperature in the oven may exceed 1650 oC, which can lead to melting of the refractory lining and complete failure of the furnace.
Loading lightweight scrap
Loading of lightweight aluminium scrap directly into the melt of reverberatory furnaces of the bath type can be a source of an additional increased increase in melting losses of metal. It happens, if dross, which formed on the surface of the melt, then exposed directly to the flame of the burners. This melting dross is a “liquid” mixture of oxides and metal entrained in the dross.. It can contain up to 80 or even 90 % good metal aluminium. If this dross is not quickly removed from the surface or treated with a flux to separate the metal from oxides and contaminants, then this metal, will quickly oxidize and this will lead to additional loss of usable metal.
Melt stirring
To reduce the formation of dross on the surface of the aluminium melt, it is important not only to carefully control the operations of loading the charge and control the temperature., but also creating conditions for better heat transfer to the melt. With a decrease in the surface temperature of the melt, the temperature difference between the heat source and the melt increases, which provides a higher rate of heat transfer and, Consequently, faster melting. This is facilitated by: 1) the correct gas/air ratio on the burners and 2) stirring the melt. This makes it possible for the heat from the radiant energy of the vault and walls to more efficiently get to the surface of the melt and spread into its depth.
Dross removal
Dry hearth furnaces are free of some of these problems., since surface oxides from the charge remain on the inclined hearth, and the molten aluminium flows into the furnace. If wet or oiled aluminium scrap is loaded onto an inclined bed, then it doesn’t create those problems, which arise when it is directly loaded into the melt. However, oxides build up quickly on the inclined hearth, which can impede the flow of molten aluminium. Therefore, these oxide accumulations need to be cleaned off regularly.
Recommended practice for the removal of “wet” dross from the melt [1]:
- at least once every four hours
- when the dross reaches its thickness 40 mm (see Figure 2).
Furnace fluxing
Dross treatment with flux
When dross forms on the surface of the melt, then it must be removed, to:
- ensure normal melting rates
- avoid further metal loss.
Methods, which are used in industry, usually is a combination of dross treatment in a furnace, as well as regular cleaning of the walls and bottom of the ovens.
For this purpose, several types of solid fluxes (powder or granular) are used. This fuxes are mixtures of chemical compounds, having different functions. These mixtures may include reactive components, as well as fillers, which provide a convenient volume or cost reduction.
Drossing fluxes
It is known, that a large percentage of usable metal may be lost, when the “wet” dross is discharged from the furnace. Wet dross contains 60-80 % of metal. Many industries are trying to reduce the metal content in such dross by treating the dross in a furnace using exothermic fluxes. These fluxes contain oxidants and fluorides, to raise the temperature and help separate the oxides from the metal. This exothermic reaction consumes aluminum to generate heat and can cause up to 20 % metal, which is contained in the dross.
Special drossing fluxes are designed to help separate metal aluminum, which is trapped in the dross. This dross consists of droplets of metallic aluminum., which is located inside the shell made of aluminum oxide (Figure 5).
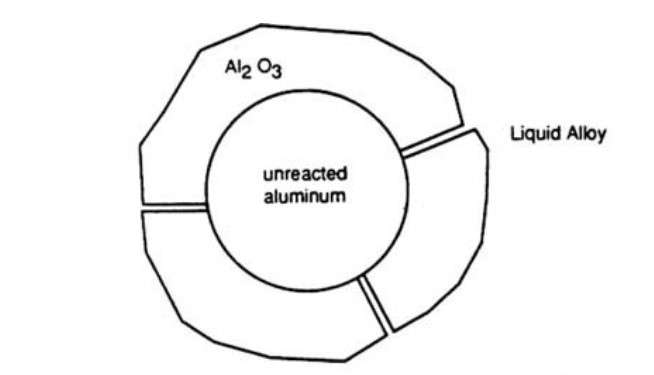 Figure 5 – Schematic view of a dross shell with aluminum trapped inside it [1]
Figure 5 – Schematic view of a dross shell with aluminum trapped inside it [1]
The composition and structure of the dross depends on the type of aluminium charge and the type of melting furnace. The fluxes used are usually reactive, which provides good contact with the dross to separate it from the aluminium. Flux can also contain exothermic components, so that the generated heat accelerates the separation process. The use of thermal and mechanical stirring results in an increase in local surface temperature and fluidity. It helps to soften the oxide shell, release of aluminum metal droplets, their coalescence and return to the melt.
Typical Aluminum Dross Fluxing Procedure
Application methods for both types of flux can vary greatly from production to production, but they all require tight mixing of the flux and dross, to achieve good separation of aluminum from oxides [1].
- The initial stage is the uniform distribution of the flux over the slag surface. Flux consumption can be different, but some flux manufacturers recommend starting around 1 kg per square meter of melt area.
- Then the flux and dross are mixed with a scraper, to start the process of separation of oxides and aluminum.
- After that, the furnace window is closed and the burners are turned on for a short time at normal power. Raising the temperature promotes the reaction of the flux with the dross and accelerates the separation of aluminum and oxide.
- The oven window is opened slightly and the operator mixes gently, rakes and breaks the slag layer with a scraper, what to even more mix flux with dross.
- This mixture is then removed from the oven so quickly, how is it possible, to reduce the heat loss of the furnace, which can be quite significant in cleaning and fluxing operations.
- You need to try, so that the aluminum freed from the dross flows as much as possible from the dross-flux mixture into the melt.
A typical such operation is shown in Figure 6.
 Figure 6 – Typical Aluminum Dross Fluxing Procedure [1]
Figure 6 – Typical Aluminum Dross Fluxing Procedure [1]
Not chemistry, but physics
Need to understand, that during the fluxing of the dross, there is no reduction of aluminum from oxide. This can only be done electrolytically. As a result of the treatment of the dross with a flux, a purely physical separation or fracture of the fragile oxide layer occurs and the release of aluminum, which was contained inside it (Figure 7).
Figure 7 – Scheme of the release of aluminium from the dross shell [1]
The purpose of fluxing dross is to turn white, lump dross with a high aluminum content, into a dark powdery dross with a low aluminum content:
- The aluminium melting dross with a high aluminum content looks light and shiny, that is, obviously saturated with aluminium (Figure 8).
- The treated aluminium melting dross looks dull and powdery (Figure 9).

Figure 8 – Untreated white dross with high aluminium content [1]
 Figure 9 – Flux treated dark powdery dross with low aluminium content [1]
Figure 9 – Flux treated dark powdery dross with low aluminium content [1]
Dross economics
- Typically, the aluminum content in the untreated dross ranges from 85 to 90 %. Furnace fluxing can return about half of this amount.
- The flux treated and skimmed dross is still suitable for additional processing on auxiliary equipment, for example, on a special crushing press. After this treatment, the amount of remaining aluminum in the dross can be about 25 % (Figure 10).
Figure 10 – Typical aluminium content in melting aluminium dross [1]
The practice of handling aluminum dross varies greatly from production to production. Some industries perform fluxing and refining operations very carefully. Others simply skim the dross into containers and send it to local slag recyclers.. These drosses account for a large proportion of metal losses, that occur in the metallurgical smelting industry. These losses can amount to 1-2 % from the weight of a clean and dry aluminium scrap. They can go up to 6-10 % and even higher for contaminated aluminium scrap.
Most efficient aluminum smelters ship dross for recycling containing aluminum 40-50 %. Those enterprises, which do not produce dross fluxing in the furnace, can ship dross containing aluminum 75 and even 85 %.
How to reduce the amount of dross
To reduce the amount of dross, which is formed in melting operations, it is recommended to adhere to following rules [1].
- Apply clean, dry aluminium scrap charge.
- Use charge materials with a high mass-to-surface ratio, to minimize the amount of oxides, which are introduced into the furnace.
- Apply coating fluxes to protect the aluminium melt from oxidation.
- Minimize the melting cycle by stirring the melt.
- Hold, as much as possible, closed furnace to reduce oxidation, especially when holding the melt.
- Avoid direct contact of the burner flame on molten aluminium.
- Ensure the correct fuel/air ratio on the burners, to minimize the conditions for oxidation.
- Hold molten metal at the lowest possible temperature.
- Minimize the transfer of molten metal through trough and ladles. Minimize turbulence and cascading when handling liquid metal.
- Apply a system for the recovery and extraction of aluminum from dross.
- Cover furnace tools with an appropriate protective coating, which is not wetted in liquid aluminium.
- Follow, so that the loading window of the furnace is hermetically closed.
- Provide overpressure in the reverberatory melting furnace.
- Follow, that the thermocouples are operational and correctly calibrated, to reliably control the temperature of the aluminum melt.
The sources:
1. Dross, Melt Loss, and Fluxing of Light Alloy Metals // ASM Handbook, Volume 15: Casting – 2008
2. Reduction of Oxidative Melt Loss of Aluminum and Its Alloys – Final Report DE-FC36-00ID13898 – DasSecat, Inc, 2006
3. Aluminum Recycling – Second Edition / Mark E. Schlesinger




